SPECTra radiopharmacy LIMS helps to ensure compliance
26/09/2019
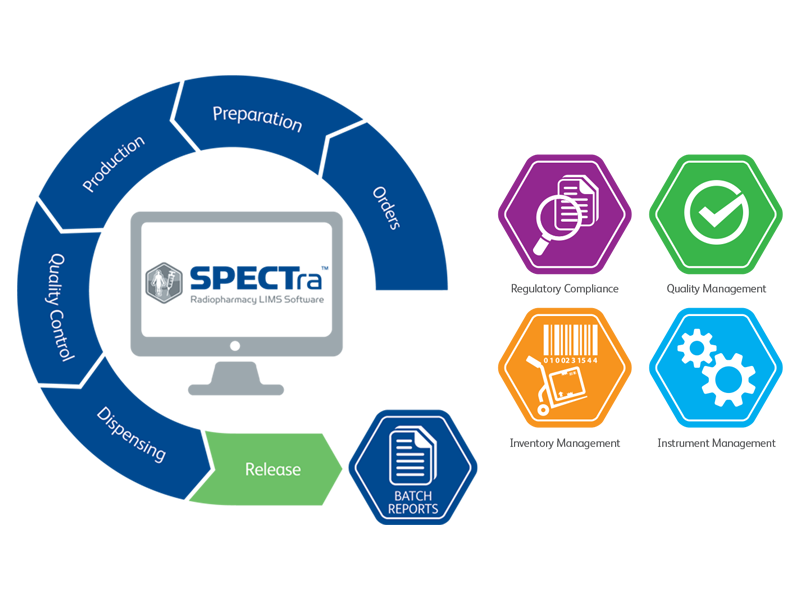
SPECTra radiopharmacy LIMS helps to ensure compliance
Personnel training records and competency against SOPs can be maintained within SPECTra, with access rights linked to the individual's training and qualifications.
SPECTra's Corrective Action and Preventive Action (CAPA) processes help you to solve problems quickly, minimise the impact of discrepancies, and reduce the chance of reoccurrence.
SPECTra can be used as a central repository for QC/QA by capturing data directly from the QC instruments used within SPECT production, feeding these results directly into the batch report.