Automating radionuclide separation at the National Physical Laboratory
26/01/2023

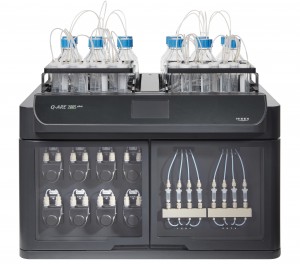
The Hidex Q-ARE 50 has two reagent zones that holds up to twelve one-liter reagent bottles, which has allowed NPL to investigate high-volume separations for radionuclides with low-activity in drinking water.
The Hidex Q-ARE 50 is an automated radionuclide extraction system that has been installed at NPL’s Nuclear Metrology Group
The first Hidex Q-ARE 50 automated radionuclide extraction system in the UK has been delivered to the National Physical Laboratory’s Nuclear Metrology Group, where it will be used to perform liquid chromatography for radionuclide separation. NPL is the UK’s National Metrology Institute and is responsible for realising, maintaining, and developing the UK’s primary measurement standards and uses them to provide standards, calibrations, measurements, and testing to end users.
Science Area Leader Ben Russell and Higher Research Scientists Hibaaq Mohamud and Alexandre Tribolet are hoping to achieve automated radionuclide separation using the Q-ARE 50 to replace existing Dry Column Vacuum Chromatography (DCVC). The Q-ARE 50 has a capacity for four samples to be processed simultaneously and two reagent zones that hold up to twelve reagent bottles.
Demonstrating reproducibility and robust methodology
Hibaaq said, “Our main interest in the Q-ARE is in automating our existing manual methods for separation where we rely on a vacuum box. In previous work, we’ve needed to demonstrate reproducibility and robust methodology, which is something the Q-ARE will allow us to improve.
“What we’ve sometimes found in previous separations using the vacuum box is variations in results, despite using a repeated methodology. There is increasing demand in nuclear medicine to use automated methods and the Q-ARE’s automation will allow us to handle high-activity materials without having to manually separate using the vacuum box.”
Elaborating on Hibaaq’s point, Alexandre said, “The vacuum box requires us to be there in person, so being able to transfer the existing separation methodology into an automated process will reduce the time taken to perform liquid chromatography and allow us to control the flow rates a lot better.
“The Q-ARE gives us a higher throughput of samples and being able to streamline that process of separation and analysis, as well as potentially coupling this with measurement by mass spectrometry to further automate our procedures, is really interesting.”
Ben said, “When performing separation for nuclear medicine, we have to very closely control the reagents and volumes that we use. For example, when we’re looking at the lanthanide series of elements, they have very similar chemical properties to each other and are challenging to cleanly separate. We can separate using vacuum boxes, but it’s a time and labour-intensive process that requires us to stay with it the entire time and track it, weighing every sample to check that volumes and flow rates are right. If we can reliably automate this process, it’s a lot more efficient use of the analysts’ time.
“As another example, we’ve been looking at high-volume separations for radionuclides with low-activity in drinking water, which means passing up to a liter through a single pump. Before the Q-ARE, we didn’t have the means to load one liter and pump that through a resin. We would have to keep going back and topping it up. That would no longer be the case.”
Automated tandem liquid chromatography
The Q-ARE has both a single and a tandem chromatography mode. Tandem chromatography allows samples and reagents to flow consecutively through two columns, with elution from each column automatically performed into two different elution bottles. Up to five elution fractions can be collected from one sample.
Alex said, “When using DCVC, we have to prepare the columns or swap them, but the Q-ARE’s tandem chromatography allows us separate two different columns and collect the elements from them. By allowing us to carry out more separations, it frees the analysts.”
Developing separation procedures using decay counting and mass spectrometry
Ben explained the diverse areas that the Nuclear Metrology Group will use the Q-ARE in.
“We develop separation procedures for subsequent measurements using decay counting or mass spectrometry for applications such as nuclear medicine, including diagnoses and therapies for cancer treatment, nuclear energy, environmental monitoring including nuclear decommissioning, and forensic analysis. This could be used for identifying isotopic ratios for determining the source of nuclear contamination following a radiological incident, or long-term monitoring of radionuclides to record ocean circulation or climate reconstruction.”
Optimising working methods
The Nuclear Metrology Group was given training by Hidex’s Product Line Manager Ari Lehmusvuori and LabLogic’s Service Operations Manager Chris Harrison prior to, during, and after installation, to allow the team to optimise their methods.
Ben recalled, “At the International Symposium on Nuclear and Environmental Radiochemical Analysis in York in September 2022, Ari gave us a demonstration of the Q-ARE after we’d ordered it but before it had been delivered. He took time out of the conference to familiarise us with it, and that was very useful.”
Hibaaq said, “Chris gave a very nice introduction to the Q-ARE and its software. He ran through a lot of different setups and we’ve since approached him about a few of the protocols we would like to try, and he was kind enough to go through each step with us.”
Alexandre added, “Chris was really approachable and easy to ask questions to. He answered everything and sent us any material that we asked for. He was very understanding with the software, which we have to screen for cybersecurity purposes as its external, and he was happy to coordinate with our IT group for that.”
Find out more
You can learn more about the Hidex Q-ARE by clicking the button below to download a product catalogue or speak to a product specialist.