How do you prove data integrity with new hardware?
06/05/2026

The answer lies in robust software OQ supported by documented evidence in line with internationally recognised requirements
In regulated environments, data integrity is fundamental to GMP compliance. Yet one part which is often overlooked is software validation when implementing new hardware. FDA 21 CFR Part 11 and EU GMP Annex 11, Chapter 4 expectations states that computerised systems must be validated, and that data must be protected from creation through review, retention, and retrieval.
So how do you truly prove data integrity?
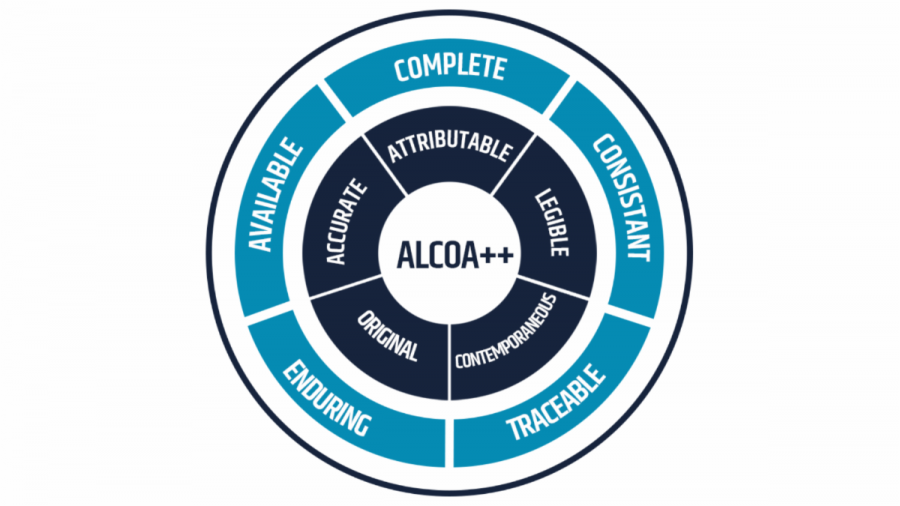
Producing data compliant with ALCOA++
Hardware qualification is essential but represents only one component of compliance. Software Installation Qualification (IQ) and Operational Qualification (OQ) are critical in demonstrating that a system operates as intended and consistently produces data that meets principles defined by ALCOA++ including traceability, security, and controlled access.
Many instrument manufacturers focus almost exclusively on hardware OQ. When software OQ is absent or limited, key system functions remain unverified, creating a compliance gap against the expectations set out in regulations such as FDA 21 CFR Part 11 and EU GMP Annex 11, Chapter 4.
LabLogic’s approach to combined validation
LabLogic takes a fundamentally different approach. Originally founded as a software company before migrating into hardware, validation principles are embedded into our systems from inception. A genuine software OQ must demonstrate that every executable action has been tested and performs as expected under defined conditions.
This approach aligns closely with expectations set by FDA 21 CFR Part 11 and EU GMP Annex 11, Chapter 4, which requires that software is fit for intended use and capable of maintaining data integrity under normal operating conditions. LabLogic therefore applies equal importance to software and hardware OQ, ensuring that system functionality, user access controls, audit trails, and data handling are fully tested and documented.
A growing regulatory expectation for software validation
Around the world, regulators are increasingly requesting explicit software validation evidence during inspections. Inspectors are no longer satisfied with confirmation that a system was installed. They expect documented evidence that software functions have been tested, verified, and shown to protect data integrity during routine use.
Challenges for end users
Not all sites have dedicated QA departments, and as a result, expectations around GMP compliance may not always be fully understood. A common misconception is that hardware OQ alone satisfies GMP validation expectations despite FDA 21 CFR Part 11 and EU GMP Annex 11, Chapter 4 clearly extending validation requirements to software.
This issue goes beyond commercial manufacturing. Research facilities are increasingly expected to demonstrate compliance, particularly when data is intended for publication or regulatory submission.
A combined validation for software and hardware
LabLogic provides hardware and software IQ/OQ services together to deliver a comprehensive, validation package from a single supplier. Our validations are thorough, structured, and designed to meet modern GMP and data integrity expectations.
By validating both hardware and software together, LabLogic helps ensure compliance and gives facilities confidence that their data truly stands up to regulatory scrutiny.
Find out more
If you’d like to know more about our qualification and validation services, click the button below to speak to a product specialist directly.